What You Missed in 2025:
Spanning the full spectrum of analytical methods, from bioassays and molecular biology to physicochemical characterization, our comprehensive 2-track agenda delivers the technical depth and practical insights you need to move your programs forward.
Biotech, pharma, and leading service providers will present data-driven, case study-led sessions on analytical innovations for AAV-based gene therapies and the evolving landscape of novel capsids, LNP characterization, and gene transfer platforms. Whether you’re refining existing assays or developing analytics for new modalities, you’ll access comprehensive, in-depth, and current expertise to help shape the next generation of gene therapy products in 2025 and beyond.

What to Expect?
Validate
Validate potency assays and genome integrity analytics across AAV, LNP, and gene editing platforms to ensure consistency, safety, and regulatory acceptance
Overcome
Conquer challenges in full/empty capsid quantification, aggregation profiling, and stability assay design, enhancing product quality, reducing batch failures
Advance
Develop analytical tools like long-read sequencing, mass photometry, AUC, and dPCR into GMP-ready QC use for precise, high-resolution measurements, streamlining lot release and increasing confidence in product integrity
Align
Coordinate assay strategies with global regulatory expectations for IND, BLA, and RMAT pathways, facilitating faster and more predictable regulatory approvals, and expediting time to clinic and market
Tackle
Address immature or under-validated techniques still holding back next-gen programs, accelerating innovation, and enabling successful translation of novel therapies into clinical pipelines
What Was New for 2025?
Garner insights into potency assay design across modalities with case studies from Sanofi, Atsena Therapeutics, Regeneron, and more, uncovering how to build functionally relevant assays for AAV, LNP, mRNA, and gene-editing platforms.
Elevate regulatory preparedness by learning from Bristol Myers Squibb, Astellas, Spark Therapeutics, and others on navigating evolving FDA and EMA expectations for genome integrity, RC virus detection, and adventitious agent testing.
Navigate analytical bottlenecks such as full/empty capsid quantification, assay variability, and method transfer with practical guidance from REGENXBIO, Adverum Biotechnologies, and Eli Lilly on leveraging AUC, dPCR, and mass photometry.
Envision future-ready analytical strategies by benchmarking against innovations from Novartis, CSL, Sarepta, and more, addressing NGS method validation, stability testing, and platform-specific challenges beyond traditional AAV-based systems.
Who Will You Meet?
Join a focused community of analytical, assay, and process development leaders from the world’s top biotech and pharma companies. You'll connect with scientists, technical directors, and QC specialists who are driving innovation in gene therapy analytics–from molecular biology and bioassays to physicochemical methods and CMC strategy. Whether you're looking to benchmark your approach, exchange real-world experiences, or forge new collaborations, this is the place to meet the minds shaping the future of analytical development.
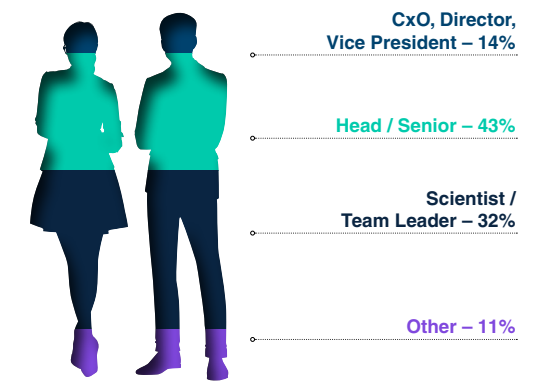
Seniority of Attendees*
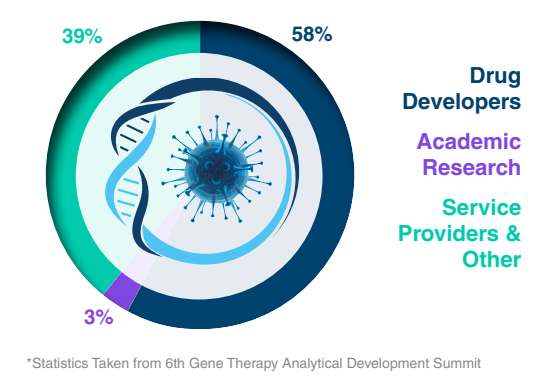
Types of Companies Attending*

This summit is a must-attend event to learn how colleagues in the field are overcoming analytical challenges and to learn about new technology applications for AAV-based gene therapy products and processes
Margaret Butko, Director CMC Assay Development, Adverum Biotechnologies
Attending Companies Included